 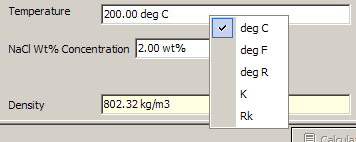
The slope essentially tells you how fast the dependent variable (y-axis) is changing when you let the independent (x-axis) vary. Engineering was established in the year 2013 with a 4 year under graduation program 1 / 7. The extra s is to account for the extra time needed to move the object that is already traveling with some velocity. Suggestions for a thermodynamics calculation program We are calculating some redox equilibria - it would be useful to see a plot of activities vs. Thermodynamics An Engineering Approach Solution Manual. And to see how long it takes, you divide by the time interval (1/s). Intuitively, you can think of it as I need to apply this force to move this object with this mass. If you think about classical mechanics, for example, a ball heading towards the earth, (neglecting air resistance and approximating the distance near the surface of the earth), the force is F=N=kg(m/s^2). This tells you how fast the pressure is changing with volume for every kg of gas/atoms in the system per volume. The thermodynamic properties of each phase are then described with a. Slope m, is equal to Pressure/Volume, or dimensionally: m=(F/A)/V in SI units, it will be kg/(m^5) or kg*(m^-4) s^(-2). CALPHAD stands for CALculation of PHAse Diagrams, a methodology introduced in 1970 by Larry. Symmetry Thermo is a set of software components used for the calculation of thermo-physical. Physical Properties Online Calculator Air - Temperature - Pressure (TP) Option Temperature Range: 273 K through 5,000 K Temperature K Air - Enthalpy. At the end of the PayPal checkout, you will be sent an email containing your key and software. The slope tells you how fast the pressure is changing with respect to volume. Ensure accurate and reliable thermodynamics properties every time. Enter your calculators 14-digit ID (F1:Tools About). But in the either case, the magnitude of the work is still the area inside the PV cycle. 
if the cycle is counterclockwise, that means the higher line (higher W area) was R to L (negative work), so the total W is negative (work was done ON the gas). The difference between the two areas is, by definition, the area within the cycle and that is the work done BY the gas during the cycle.Īs for the sign, if the cycle is clockwise, that means the higher line (higher work area) was L to R (positive work), so the total W is positive. The total work of the cycle is the sum of these two works (areas under the curves), but since one is negative, it's essentially the difference. While the cycle is moving R to L (decreasing volume) the work done BY the gas is considered negative. 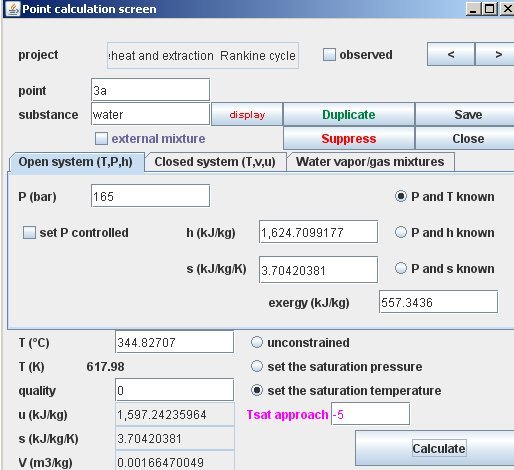
While the cycle is moving L to R (increasing volume) the work done BY the gas (area under the curve) is considered positive. Advances in first-principles calculations of thermodynamic properties of planetary materials (Invited)įirst-principles atomistic simulation is a vital tool for understanding the properties of materials at the high-pressure high-temperature conditions prevalent in giant planet interiors, but properties such as solubility and phase boundaries are dependent on entropy, a quantity not directly accessible in simulation.This is now explained in a little pop-up box in the middle of the article, but you may have missed it or it may have been added after your question. How can I calculate the work done I dont think I can apply W P d V. Determining entropic properties from atomistic simulations is a difficult problem typically requiring a time-consuming integration over molecular dynamics trajectories. The following thermodynamic properties are calculated: density water, dynamic viscosity water, kinematic viscosity water, specific inner energy water. In the entrence of the turbine and the exit we have the following properties: enthalpy, temperature, volume and interne energy. Here I will describe recent advances in first-principles thermodynamic calculations which substantially increase the simplicity and efficiency of thermodynamic integration and make entropic properties more readily accessible. I will also describe the use of first-principles thermodynamic calculations for understanding problems including core solubility in gas giants and superionic phase changes in ice giants, as well as future prospects for combining first-principles thermodynamics with planetary-scale models to help us understand the origin and consequences of compositional inhomogeneity in giant planet interiors.įORTRAN 4 computer program for calculation of thermodynamic and transport properties of complex chemical systemsĪ FORTRAN IV computer program for the calculation of the thermodynamic and transport properties of complex mixtures is described. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
